Lesson: Metals and Non-metals
Question 1
Give
an example of a metal which
(a)
is a liquid at room temperature.
(b)
can be easily cut with a knife.
(c)
is the best conductor of heat.
(d)
is a poor conductor of heat.
Solution:
(a)
Mercury
(b)
Sodium
(c)
Silver
(d)
Mercury and lead
Question 2
Explain
the meanings of malleable and ductile.
Solution:
A
substance is said to be malleable if it can be beaten into thin sheets.
A
substance is said to be ductile if it can be drawn into thin wires.
Generally
metals are both ductile and malleable.
Question 3
Why
sodium is kept immersed in kerosene oil?
Solution:
Sodium is a
highly reactive element. It is kept immersed in kerosene oil to avoid its
reaction with oxygen. Such reactions are also explosive in nature.
Keeping sodium
immersed in kerosene helps:
a) preventing accidental damage.
b) storing sodium in its pure form.
Question 4
Write
equations for the reactions of:
(i)
Iron with steam
(ii)
Calcium and potassium with water
Solution:
(i)
(ii)
Question 5
Samples
of four metals A, B, C and D were taken and added to the following solution one
by one. The results obtained have been tabulated as follows.
|
Metal
|
Iron (II) Sulphate
|
Copper (II) Sulphate
|
Zinc Sulphate
|
Silver nitrate
|
|
A
B
C
D
|
No reaction
Displacement
No reaction
No reaction
|
Displacement
No reaction
No reaction
|
No reaction
No reaction
No reaction
|
Displacement
No reaction
|
Use the Table
above to answer the following questions about metals A, B, C and D.
(i) Which
is the most reactive metal?
(ii) What
would you observe if B is added to a solution of Copper(II) sulphate?
(iii)
Arrange the metals A, B, C and D in the order of decreasing reactivity.
Solution:
(i) B is
the most reactive metal.
(ii) B will
displace copper from the copper sulphate solution.
(iii)
Arrangement of metals in the order of decreasing reactivity:
Question 6
Which
gas is produced when dilute hydrochloric acid is added to a reactive metal?
Write the chemical reaction when iron reacts with dilute .
Solution:
When a reactive metal reacts with dilute hydrochloric acid, hydrogen gas
is produced.
The chemical reaction when iron reacts with dilute is as follows:
Question 7
What
would you observe when zinc is added to a solution of iron(II) sulphate? Write
the chemical reaction that takes place?
Solution:
Zinc is more
reactive than iron. So, when it is added to iron(II) sulphate solution, it
displaces iron from the solution.
The chemical
reaction is as follows:
Question 8
(i)
Write the electron-dot structures for sodium, oxygen and magnesium.
(ii)
Show the formation of and by the transfer of electrons.
(iii)
What are the ions present in these compounds?
Solution:

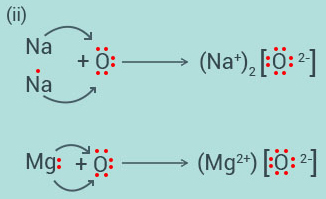
(iii)
The
ions present in
The ions present in
Question 9
Why
do ionic compounds have high melting points?
Solution:
The
force of attraction in ionic bonds is very strong. Therefore, the ionic
compounds require a lot of energy to overcome this bond. That is why ionic
compounds have high melting points.
Question 10
Define
the following terms.
(i)
Mineral
(ii)
Ore
(iii)
Gangue
Solution:
(i)
Mineral are the naturally
occurring materials in which metals or their compounds are found on earth.
(ii)
Ores are the minerals from which
metals can be extracted profitably.
(iii)
Gangues are the impurities
present in an ore.
Question 11
Name
two metals which are found in nature in the free state.
Solution:
Silver and
gold
Question 12
What
chemical process is used for obtaining a metal from its oxide?
Solution:
A metal can be obtained
from its oxide by the process of reduction.
Question 13
Metallic oxides of zinc, magnesium and copper were
heated with the following metals.
|
Metal
|
Zinc
|
Magnesium
|
Copper
|
|
Zinc oxide
|
|
|
|
|
Magnesium oxide
|
|
|
|
|
Copper oxide
|
|
|
|
In which cases will you find displacement reactions
taking place?
Solution:
|
Metal
|
Zinc
|
Magnesium
|
Copper
|
|
Zinc oxide
|
No reaction
|
Displacement
|
No reaction
|
|
Magnesium oxide
|
No reaction
|
No reaction
|
No reaction
|
|
Copper oxide
|
Displacement
|
Displacement
|
No reaction
|
Question 14
Which metals do not corrode easily?
Solution:
Metals
which are least reactive, such as silver, gold, etc. do not corrode easily.
Question 15
What
are alloys?
Solution:
An
alloy is a homogeneous mixture of two or more metals, or a metal and a
non-metal. The properties of an alloy are different from the constituent
matters.

