Lesson: Carbon and its Compounds
Question: 1
Ethane, with the molecular formula has:
(a) 6 covalent bonds.
(b) 7 covalent bonds.
(c) 8 covalent bonds.
(d) 9 covalent bonds.
Solution:
b
Question:2
Butanone is a four-carbon compound with
the functional group:
(a) Carboxylic acid
(b) Aldehyde
(c) Ketone
(d) Alcohol
Solution:
c
Question:3
While cooking, if the bottom of the
vessel is getting blackened on the outside, it means that:
(a) The food is not cooked completely.
(b) The fuel is not burning completely.
(c) The fuel is wet.
(d) The fuel is burning completely.
Solution:
b
Question:4
Explain the nature of the covalent bond
using the bond formation in .
Solution:
The structure of is given
below.
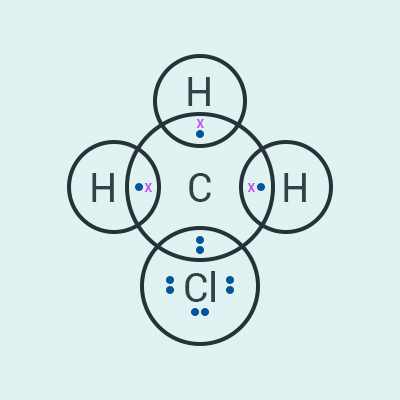
Carbon has four valence electrons. In order to form
an octet, it needs four more electrons. It shares 1 electron each with 3
hydrogen atoms and 1 electron with chlorine atom. In this way, chlorine and the
three hydrogen atoms attain a stable configuration. Since these bonds are
formed because of sharing of electrons, these are therefore called covalent
bonds.
Question:5
Draw the electron dot structures for:
(a) Ethanoic acid
(b)
(c) Propanone
(d)
Solution:
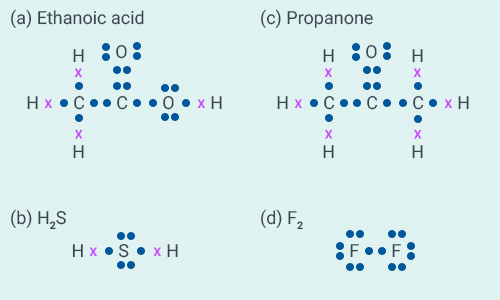
Question:6
What is a homologous series? Explain
with an example.
Solution:
A series of carbon compounds is said to be
homologous when difference between two successive compounds is .
Example: Methane, ethane, propane, butane, etc.
belong to the alkane homologous series. The general formula of this series is .
Formula of methane:
Formula of ethane:
Formula of propane:
Formula of butane:
Here, there is a difference of unit
between two successive compounds.
Question:7
How can ethanol and ethanoic acid be
differentiated on the basis of their physical and chemical properties?
Solution:
Ethanol and Ethanoic acid can be
differentiated on the basis of their following properties:
|
Ethanol
|
Ethanoic
acid
|
|
Physical Properties:
|
|
It has a pleasant smell.
|
It has a pungent vinegar-like smell.
|
|
It does not freeze in winter
|
It freezes in winter.
|
|
Chemical Properties:
|
|
It does not react with metal
carbonates.
|
It reacts with metal carbonates.
|
|
It does not react with NaOH.
|
It reacts with NaOH.
|
Question: 8
Why does micelle formation take place
when soap is added to water? Will a micelle be formed in other solvents such as
ethanol also?
Solution:
A soap molecule has two ends. One end is
hydrophilic and the other end is hydrophobic. When soap is dissolved in water
and clothes are put in the solution, soap molecules arrange themselves in a
cluster. In this, the hydrophilic end is
outside the sphere and hydrophobic end is towards the centre of the sphere.
The dirt present in the clothes is insoluble in
water. The hydrophobic ends of the clusters attach themselves to the dirt. This
cluster formation in which the dirt is entrapped is called micelle.
Micelles will not be formed in ethanol as it is not
as polar as soap.
Question:9
Why is carbon and its compounds used as
fuels for most applications?
Solution:
Carbon and its compounds are used as fuel as they have
large number of carbon and hydrogen. When burnt in the presence of air, they
give a large amount of heat as they have high calorific value. Therefore,
carbon and its compounds are used as fuels for most applications.
Question: 10
Explain the formation of scum when hard
water is treated with soap.
Solution:
Hard water contains salts of calcium and magnesium. When it
is treated with soap, the soap molecules react with these salts and form a
precipitate. This precipitate is called scum. A large amount of soap is wasted
in the formation of scum before it is used to wash clothes. This makes soap
unsuitable to wash clothes in hard water.
Question: 11
What change will you observe if you
test soap with litmus paper (red and blue)?
Solution:
Soap is basic in nature. So, it will
turn red litmus blue. However, the colour of blue litmus will remain blue.
Question: 12
What is hydrogenation? What is its
industrial application?
Solution:
The addition of hydrogen to an unsaturated
hydrocarbon to obtain saturated hydrocarbon is called hydrogenation. This
reaction is carried out in the presence of catalysts like nickel or palladium.
Industrial application:
·
In
petrochemical industries, hydrogenation is used to convert alkenes into alkanes
(paraffins) and cycloalkanes.
·
It
is also used to prepare vegetable ghee from vegetable oils.
Question: 13
Which of the following hydrocarbons
undergo addition reactions?
.
Solution:
are
unsaturated hydrocarbons. Unsaturated hydrocarbons undergo addition reactions.
Question: 14
Give a test that can be used to
differentiate chemically between butter and cooking oil.
Solution:
Butter contains saturated fat while
cooking oil has unsaturated fat. All unsaturated carbon compounds decolorize
bromine water whereas saturated hydrocarbons do not decolorize bromine water.
So, bromine water test can be applied to differentiate between butter and
cooking oil.
Question: 15
Explain the mechanism of the cleaning
action of soaps.
Solution:
Soap molecules have two ends. One end is
hydrophilic and another end is hydrophobic. When soap is dissolved in water,
the soap molecules arrange themselves in a cluster. Since the dirt present in
the clothes is insoluble in water, the hydrophobic ends of the clusters attach
themselves to the dirt. The dirt is entrapped in the cluster. This cluster
formation in which the dirt is entrapped is called micelle. These micelles
remain suspended in the water. When rinsed, the dirt particles are washed away
with water.

