Lesson:Metals and Non-metals
Question 1
Which
of the following pairs will give displacement reactions?
(a)
solution and copper metal
(b)
solution and aluminium metal
(c)
solution and silver metal
(d)
solution and copper metal.
Solution:
(d)
Question 2
Which
of the following methods is suitable for preventing an iron frying pan from
rusting?
(a)
Applying grease
(b)
Applying paint
(c)
Applying a coating of zinc
(d)
All of the above.
Solution:
(c)
Question 3
An
element reacts with oxygen to give a compound with a high melting point. This
compound is also soluble in water. The element is likely to be
(a)
Calcium
(b)
Carbon
(c)
Silicon
(d)
Iron.
Solution:
(a)
Question 4
Food
cans are coated with tin and not with zinc because
(a)
Zinc is costlier than tin.
(b)
Zinc has a higher melting point than tin.
(c)
Zinc is more reactive than tin.
(d)
Zinc is less reactive than tin.
Solution:
(c)
Question 5
You
are given a hammer, a battery, a bulb, wires and a switch.
(a) How could
you use them to distinguish between samples of metals and non-metals?
(b) Assess the
usefulness of these tests in distinguishing between metals and non-metals.
Solution:
(a) If the given sample can be beaten into thin sheets by the hammer, then
it is a metal, otherwise it is a non-metal.
Similarly, if a circuit made by the given sample, the
battery, the bulb, the wires and the switch conducts electricity, then it is a
metal. Otherwise the sample is a non-metal.
(b) The above tests are useful as they help in distinguishing between metals and non-metals
without involving any chemical reaction.
Question 6
What
are amphoteric oxides? Give two examples of amphoteric oxides.
Solution:
The
oxides which behave both as acidic as well as basic in nature are called
amphoteric oxides.
Examples:
aluminium oxide ( ),
zinc oxide ( )
Question 7
Name
two metals which will displace hydrogen from dilute acids, and two metals which
will not.
Solution:
Sodium and aluminium will displace hydrogen from
dilute acids.
Mercury and copper cannot displace hydrogen from
dilute acids.
Question 8
In
the electrolytic refining of a metal M, what would you take as the anode, the
cathode and the electrolyte?
Solution:
In the electrolytic refining of a
metal M:
·
The anode is the impure metal M
·
The cathode is the thin strip of
pure metal M
·
The electrolyte is the solution of
the salt of the metal M
Question 9
Pratyush
took sulphur powder on a spatula and heated it. He collected the gas evolved by
inverting a test tube over it, as shown in figure below.
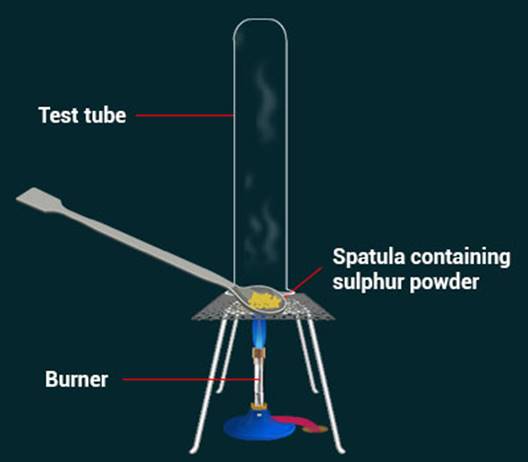
(a)
What will be the action of gas on?
(i) Dry litmus paper?
(ii) Moist litmus paper?
(b)
Write a balanced chemical equation for the reaction taking place.
Solution:
(a) (i) There will be no action on the dry
litmus paper.
(ii) The colour
of the moist litmus paper turns red as sulphur is a non-metal and the oxides of
non-metals are acidic in nature.
(b)
Question 10
State
two ways to prevent the rusting of iron.
Solution:
Two
ways to prevent the rusting of iron are:
(i)
Application of oil or paint
(ii)
Galvanisation
Question 11
What types of oxides are formed when non-metals
combine with oxygen?
Solution:
Neutral or acidic oxides are formed when non-metals
combine with oxygen. Examples are and are neutral oxides.
Question 12
Give reasons:
a) Platinum, gold and silver are used to make
jewellery.
b) Sodium, potassium and lithium are stored under oil.
c) Aluminium is a highly reactive metal, yet it is
used to make utensils for cooking.
d) Carbonate and sulphide ores are usually converted
into oxides during the process of extraction.
Solution:
(a) Platinum, gold and silver are used to make
jewellery because they are very lustrous, less reactive and do not corrode
easily.
(b) Sodium, potassium and lithium, being highly
reactive, are stored under oil to prevent their reaction with air and moisture.
(c) Aluminium is light in weight and is a good
conductor of heat. It reacts with oxygen present in the air to form a thin
layer of aluminium oxide. This oxide layer is very stable. It prevents further
reaction of aluminium with oxygen.
So, even
though aluminium is a highly reactive metal, yet it is used
to make utensils for cooking.
(d) Carbonate and sulphide ores are usually converted
into oxides during the process of extraction. This is due to the fact that
metals can be more easily extracted from their oxides rather than from their
carbonates and sulphides.
Question 13
You
must have seen tarnished copper vessels being cleaned with lemon or tamarind
juice. Explain why these sour substances are effective in cleaning the vessels.
Solution:
Copper
reacts with carbon dioxide in the air to form copper carbonate. Due to this, it
loses its shiny brown surface and a green coating of copper carbonate is formed.
The citric acid present in the lemon or tamarind neutralises the copper
carbonate, thereby dissolving the layer and thus helping in cleaning the
vessel.
Question 14
Differentiate between metal and non-metal on the basis of their chemical
properties.
Solution:
|
Metals |
Non-metals |
|
Metals form basic
oxides on reaction with oxygen. |
Non-metals form acidic
oxides or neutral oxides on reaction with oxygen. |
|
Some metals displace
hydrogen from water. |
Non-metals do not react
with water to evolve hydrogen. |
|
Metals usually displace
hydrogen from dilute acids. |
Non-metals do not react
with dilute acids. |
Question 15
A man went door to door posing as a goldsmith. He promised to bring back
the glitter of old and dull gold ornaments. An unsuspecting lady gave a set of
gold bangles to him which he dipped in a particular solution. The bangles
sparkled like new but their weight was reduced drastically. The lady was upset
but after a futile argument the man beat a hasty retreat. Can you play the
detective to find out the nature of the solution he had used?
Solution:
The solution he
had used was Aqua regia. It is a mixture of concentrated hydrochloric acid and
concentrated nitric acid in the ratio 3:1. When a gold ornament is cleaned with
Aqua regia, some of the gold gets dissolved in it. This results in the loss in
weight of the gold ornament.
Question 16
Give reasons
why copper is used to make hot water tanks and not steel (an alloy of iron).
Solution:
Copper does not
react with any form of water, be it cold, hot or steam. On the other hand, steel
(an alloy of iron) reacts with steam to form iron oxide. Due to this reason,
copper is used to make hot water tanks, and not steel.