Lesson: Combustion and Flame Science
Exercise:
List conditions under which combustion can take place.
Solution:
The conditions required for combustion to take place are:
· Presence of a fuel
· Reaching the ignition temperature (i.e., the minimum temperature at which a substance catches fire)
· Presence of air (i.e., oxygen)
Question: 2
Fill in the blanks:
(a) Burning of wood and coal causes _________of air.
(b) A liquid fuel used in homes is ___________.
(c) Fuel must be heated to its ___________ before it starts
burning.
(d) Fire produced by oil cannot be controlled by _________.
Solution:
(a)
Burning of wood and coal causes pollution of air.
(b) A liquid fuel used in homes is LPG.
(c) Fuel must be heated to its ignition temperature before it
starts burning.
(d) Fire produced by oil cannot be controlled by water.
Question: 3
Explain how the use of CNG in automobiles has reduced pollution in our cities.
Solution:
Combustion of fuels like petroleum causes formation of harmful un-burnt carbon particles along with the release of carbon monoxide gas. These pollutants enter the air and may further lead to respiratory diseases. Compressed Natural Gas (CNG) produces harmful products in much smaller quantities than other fuels, and it is comparatively a cleaner fuel. Therefore, the use of CNG can reduce pollution.
Question: 4
Compare LPG and wood as fuels.
Solution:
|
LPG |
Wood |
|
It has a calorific value of around 55000 kJ/kg. |
It has a calorific value of around 17000 to 22000 kJ/kg. |
|
It does not have a negative impact on the environment. |
On burning, it releases unburnt carbon particles, which may cause diseases. |
|
It is a smokeless fuel. |
It gives out lot of smoke. |
|
It is easy to store and can be transported. |
It needs a lot of space to store and is difficult to transport. |
|
It has low ignition temperature. |
It has high ignition temperature. |
Question: 5
Give reasons.
(a) Water is not used to control fires involving electrical equipment.
(b) LPG is a better domestic fuel than wood.
(c) Paper by itself catches fire easily whereas a piece of paper wrapped around an aluminium pipe does not.
Solution:
(a) Water is a good conductor of electricity, and so it can easily conduct electric current and cause danger of electric shocks or short-circuits. Therefore, water is unsuitable for controlling fires from electrical equipments.
(b) LPG is a better domestic fuel than wood because it does not produce smoke or release un-burnt carbon particles that cause respiratory problems.
(c) Paper has low ignition temperature and so it can by itself catch fire easily but when wrapped around an aluminium pipe, its temperature is lowered because the aluminium metal readily absorbs the heat supplied to the paper. So, the paper does not catch fire.
Question: 6
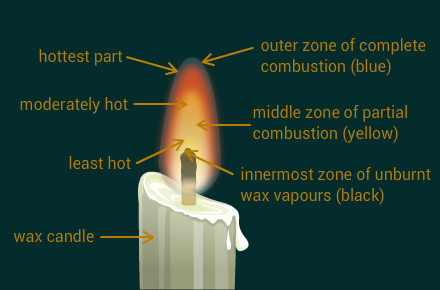
Make a labelled diagram of a candle flame.
Solution:
Question: 7
Name the unit in which the calorific value of a fuel is expressed.
Solution:
The calorific value of a fuel is expressed in kilojoules per kilogram (kJ/kg).
Question: 8
Explain how CO2 is able to control fires.
Solution:
Since CO2 is heavier than oxygen, it covers the fire like a blanket. Therefore, the contact between the fuel and oxygen is cut off, and this way, the fire is controlled.
Question: 9
It is difficult to burn a heap of green leaves but dry leaves catch fire easily. Explain.
Solution:
Green leaves have a lot of moisture in them. This moisture does not allow the leaves to catch fire easily. Dry leaves do not have any moisture. Therefore, they catch fire easily.
Question: 10
Which zone of a flame does a goldsmith use for melting gold and silver and why?
Solution:
The outer part of the candle flame is used by the goldsmith for melting gold and silver. This is due to the fact that in this part, the temperature is the highest. The high temperature helps to melt these metals easily.
Question: 11
In an experiment 4.5 kg of a fuel was completely burnt. The heat produced was measured to be 180,000 kJ. Calculate the calorific value of the fuel.
Solution:
The
amount of heat produced by the complete combustion of 1 kg of fuel is called
the calorific value of the fuel.
Given,
Heat
produced by 4.5 kg of fuel = 180000 kJ
Therefore,
the heat produced by 1kg of fuel
Hence, the
calorific value of the fuel is 40,000 kJ/kg.
Question: 12
Can the process of rusting be called combustion? Discuss.
Solution:
Combustion is a chemical process. In this process, a substance reacts with oxygen and gives out energy, in the form of either heat or light, or both.
Rusting of iron is an exothermic reaction. As heat is released during rusting, it can be called combustion.
Question: 13
Abida and Ramesh were doing an experiment in which water was to be heated in a beaker. Abida kept the beaker near the wick in the yellow part of the candle flame. Ramesh kept the beaker in the outermost part of the flame. Whose water will get heated in a shorter time?
Solution:
The water in Ramesh's beaker will heat up in a shorter time because the outermost zone of a flame is the hottest. Abida has kept the beaker in the yellow zone. This zone is comparatively less hot.