Lesson: Materials: Metals and Non-Metal
Exemplar
Which of the following is not a metal?
(a) Copper
(b) Sulphur
(c) Aluminium
(d) Iron
Solution:
b
Question: 2
The substance that will be flattened on beating with a hammer is:
(a) Crystal of iodine
(b) Lump of sulphur
(c) Piece of coal
(d) Zinc granule
Solution:
d
Question: 3
Boojho has learnt that non-metals on beating with a hammer are generally broken into pieces.
Which of the following is a non-metal?
(a) Iron nail
(b) Aluminium wire
(c) Copper plate
(d) Piece of coal
Solution:
d
Question: 4
Materials which can be drawn into wires are called ductile. Which of the following is not a ductile material?
(a) Silver
(b) Copper
(c) Sulphur
(d) Aluminium
Solution:
c
Question: 5
Metals are generally hard. Which of the following metals is an exception and can be cut with a knife?
(a) Iron
(b) Sodium
(c) Gold
(d) Magnesium
Solution:
b
Question: 6
Metals are generally solid. Which of the following metals is in the liquid state at room temperature?
(a) Mercury
(b) Silver
(c) Aluminium
(d) Sodium
Solution:
a
Question: 7
Metals generally react with dilute acids to produce hydrogen gas. Which one of the following metals does not react with dilute hydrochloric acid?
(a) Magnesium
(c) Iron
(b) Aluminium
(d) Copper
Solution:
d
Question: 8
Which of the following reacts with cold water vigorously?
(a) Carbon
(b) Sodium
(c) Magnesium
(d) Sulphur
Solution:
b
Question: 9
The metal which produces hydrogen gas on reaction with dilute hydrochloric acid as well as sodium hydroxide solution is:
(a) Copper
(b) Iron
(c) Aluminium
(d) Sodium
Solution:
c
Question: 10
Which of the following non-metals reacts and catches fire on exposure to air?
(a) Phosphorus
(b) Nitrogen
(c) Sulphur
(d) Hydrogen
Solution:
a
Question: 11
Generally metallic oxides are basic and non-metallic oxides are acidic in nature. Solution of which of the following oxides in water will change the colour of blue litmus to red?
(a) Sulphur dioxide
(b) Magnesium oxide
(c) Iron oxide
(d) Copper oxide
Solution:
a
Question: 12
Which of the following property is not responsible for copper to be used as electrical conduction wires?
(a) Ductility
(b) Colour
(c) Good conductor of electricity
(d) It is solid
Solution:
b
VERY SHORT ANSWER QUESTIONS (8)
Question: 1
Name two soft metals which can be cut with a knife.
Solution:
(a) Sodium
(b) Potassium
Question: 2
Which non-metal is essential for our life and all living beings inhale it during breathing?
Solution:
Oxygen gas
Question: 3
Name two major non-metals which are present in fertilisers and enhance the growth of plants.
Solution:
(a) Nitrogen
(b) Phosphorus
Question: 4
Which non-metal is used to disinfect water?
Solution:
Chlorine
Question: 5
A purple coloured non-metal forms a brown solution in alcohol which is applied on wounds as an antiseptic. Name the non-metal.
Solution:
Iodine
Question: 6
Zinc sulphate forms a colourless solution in water. Will you observe any colour on adding copper turning in it?
Solution:
There will not be any colour, because displacement reaction does not take place.
Question: 7
Why are bells made of metals?
Solution:
Metals are sonorous so, bells are made of metals.
Question: 8
Which liquid metal is used for making thermometers?
Solution:
Mercury
Question: 9
Which of the following metals can displace the other two metals from their salt solutions?
zinc, iron, copper
Solution:
Zinc
SHORT ANSWER QUESTIONS (6)
Question: 1
Paheli bought a statue made of copper.
To her surprise it acquired a dull green coating after a couple of months.
Explain the reason.
Solution:
Copper reacts with moist air (water, oxygen and carbon dioxide) to form a mixture of copper hydroxide and copper carbonate. This mixture looks like a green coating on copper objects.
Question: 2
In the figure below, you find that the bulb glows when an iron nail is placed between two ends of wire.
Complete the following sentenceson the bases of this fact.
(a) __________ is a metal.
(b) Metals are good __________ of electricity.
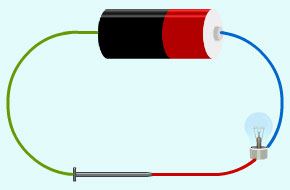
Solution:
(a) Iron
(b) Conductor
Question: 3
If, in the figure above, iron nail is replaced by a wooden stick, will the bulb glow or not? Justify your answer.
Solution:
Wood is not a good conductor of electricity, therefore the bulb will not glow.
Question: 4
Paheli prepared a blue coloured solution of copper sulphate in beaker A and placed an iron nail in it. Boojho prepared a yellowish green solution of ferrous sulphate in beaker B and placed a copperwire in it. What changes will they observe in the two beakersafter an hour?
Solution:
In beaker A, there will be a reddish brown layer of copper deposit on the iron nail and the blue coloured solution will turn yellowish green. In Beaker B, there will be no change.
Question: 5
A doctor prescribed a tablet to a patient suffering from iron deficiency. The tablet does not look like iron. Explain.
Solution:
The tablet prescribed by doctor contains iron salt and not iron metal.
Question: 6
Match the substances in Column A with their applications givenin Column B.
Column A Column B
(Substance) (Application)
(a) Oxygen (i) Making crackers
(b) Copper (ii) Disinfecting water
(c) Sulphur (iii) All living beings inhale during breathing
(d) Iron (iv) Making electric wires
(e) Chlorine (v) Making rails
Solution:
(a)-(iii), (b)-(iv), (c)-(i), (d)-(v), (e)-(ii)
(a) Oxygen (iii) All living beings inhale during breathing
(b) Copper (iv) Making electric wires
(c) Sulphur (i) Making crackers
(d) Iron (v) Making rails
(e) Chlorine (ii) Disinfecting water
LONG ANSWER QUESTIONS (5)
Question: 1
Some of the following statements are incorrect. Find the incorrect statements and correct them.
(a) The property of metals by virtue of which they can be drawn into wires is called ductility.
(b) Metals are good conductor of electricity but poor conductor of heat.
(c) Articles made of metals produce ringing sound when struck hard.
(d) Oxides of non-metals and metals are acidic in nature.
(e) A less reactive metal replaces a more reactive metal from its salt solution in water.
Solution:
Statements (b), (d) and (e) are not correct.
(b) Metals are good conductor of heat and also electricity.
(d) Oxides of non-metals are acidic in nature, but oxides of metals are basic in nature.
(e) A more reactive metal replaces a less reactive metal from its salt solution in water.
Question: 2
Iron is more reactive than copper. Can you write an activity to show this?
Solution:
When an iron nail is put in a beaker containing blue copper sulphate solution, the iron displaces copper from the solution, and a reddish brown layer of copper is deposited on the iron nail, while the blue solution turns yellowish green. The products which are obtained as a result of the chemical reaction are copper metal and iron sulphate. This shows that iron is more reactive than copper.
Question: 3
Fill in the blanks to complete the following paragraph.
The name of the product formed in the reaction of sulphur and_____________ is sulphur dioxide gas. When sulphur dioxide isdissolved in_________________, sulphurous acid is formed. The sulphurous acid turns _____________ litmus paper to ____________. Generally oxides of __________ are acidic in nature. After completing the paragraph write two questions which youcan raise on the basis of this information.
Solution:
Oxygen, water, blue, red, non-metals.
Questions may be
(a) Name the gas that is formed when sulphur reacts with oxygen.
(b) What is the nature of oxides of non-metals?
Question: 4
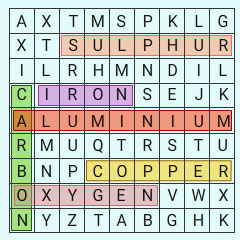
Find out the names of three metals and three non-metals from the box given as?

Solution
Metals Non-metals
Iron Carbon
Copper Oxygen
Aluminium Sulphur
Question: 5
Complete the crossword given in figure below with the help of the clues.

Across:
1. Which is generally hard, ductile, malleable and sonorous.
2. A metal is called so it can be drawn into wires.
3. Metal bells are used because of this property.
Down:
4. A metal generally used for making jewellery.
5. A metal which is liquid at room temperature.
6. A metal which reacts with acid as well as base to form hydrogen gas.
7. Substances used to enhance the growth of plants.
8. Property by virtue of which metals can be beaten into thin sheets.
Solution
